Izici
I-electrode ye-ion eku-inthanethi ilinganiswa ngesisombululo samanzi ukuhlushwa kwe-chlorine ion noma ukunqunywa komngcele kanye ne-electrode yenkomba ama-ion e-fluorine/chlorine ukuze kwakhiwe izinhlanganisela ezizinzile zokuhlushwa kwe-ion.
| Isimiso sokulinganisa | I-ion selective potentiometry |
| Ububanzi bokulinganisa | 0.0~2300mg/L |
| Izinga lokushisa elizenzakalelayoububanzi besinxephezelo | 0~99.9℃,ngo-25℃ njengobaizinga lokushisa elibhekiselwe |
| Ibanga lokushisa | 0~99.9℃ |
| Izinga lokushisa elizenzakalelayoisinxephezelo | 2.252K,10K,I-PT100,PT1000etc |
| Isampula yamanzi ihlolwe | 0~99.9℃,0.6MPa |
| Ama-ion okuphazamisa | AL3+,Fe3+,OH-njll. |
| ububanzi benani le-pH | 5.00~10.00PH |
| Amandla angenalutho | > 200mV (amanzi ahlanjululwe) |
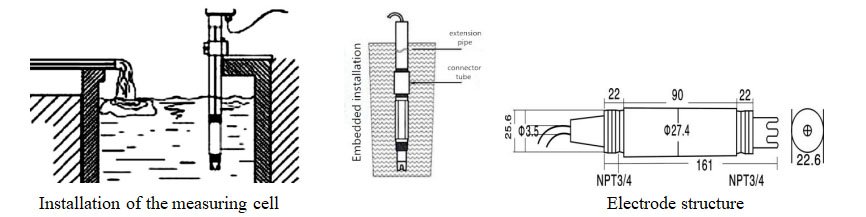
| Ubude be-electrode | 195mm |
| Izinto eziyisisekelo | I-PPS |
| Intambo ye-electrode | Intambo yepayipi engu-3/4(I-1965)I-NPT) |
| Ubude bekhebula | Amamitha ama-5 |
I-ion iyi-athomu noma i-molecule eshajiwe. Ishajiwe ngoba inani lama-electron alilingani nenani lama-proton ku-athomu noma ku-molecule. I-athomu ingathola i-chaji enhle noma i-chaji engemihle kuye ngokuthi inani lama-electron ku-athomu likhulu noma lingaphansi kunenani lama-proton ku-athomu.
Uma i-athomu ikhangwa yinye i-athomu ngoba inenani elingalingani lama-electron nama-proton, i-athomu ibizwa ngokuthi i-ION. Uma i-athomu inama-electron amaningi kunama-proton, iba yi-ion engemihle, noma i-ANION. Uma inama-proton amaningi kunama-electron, iba yi-ion engemihle.















